 Unlike in a molecule of carbon dioxide, the individual bond dipole moments in a molecule of water do not cancel out. Because the central oxygen atom bonds with two other atoms and contains two lone pairs of electrons, the structure of H 2O is bent (with a bond angle of 104.5°). In addition, the oxygen atom has a partially-negative charge, while the two hydrogen atoms have partially-positive charges. The two arrows representing the molecule’s bond dipole moments point away from the less electronegative hydrogen atoms and towards the more electronegative oxygen atom. In a molecule of water (H 2O), there are two polar covalent bonds each involves the central oxygen atom and an attached hydrogen atom. As a result, they cancel each other out, resulting in a net dipole moment of zero. In a molecule of CO 2, the two individual bond dipole moments are equal in magnitude and opposite in direction. This means that CO 2 has a net dipole moment of zero and is a non-polar molecule. Therefore, the two bond dipole moments run in opposite directions and cancel each other out. In addition, because the central atom bonds with two other atoms and lacks any lone pairs of electrons, CO 2 has a linear structure (with a bond angle of 180°). The two arrows representing the molecule’s bond dipole moments point away from the carbon atom and towards the oxygen atoms as a result. Since oxygen is more electronegative than carbon, both oxygen atoms have a partially-negative charge, while the carbon atom has a partially-positive charge. In a molecule of carbon dioxide (CO 2), there are two polar covalent bonds each involves the central carbon atom and an attached oxygen atom. Dipole Moment Examples CO 2 Dipole Moment In this case, the molecule would be classified as non-polar, despite the fact that it contains multiple polar bonds. Therefore, it is possible for the net dipole moment of a molecule to be zero when a symmetric molecule has individual bond dipole moments (equal in magnitude but opposite in direction) that cancel each other out. Īs mentioned above, dipole moments are a vector quantity because they have both a magnitude (determined by the electronegativity difference) and a direction (based on the movement of electrons toward the more electronegative atom). The SI unit for dipole moment is the coulomb-meter ( ) however, it is most commonly measured in debyes ( ). To calculate the dipole moment of a chemical bond, the following formula is used: (where is the bond dipole moment, is the magnitude of the separated charge, and is the distance of separation between the charges). In addition, the net dipole moment is affected by the geometry of the molecule, which can be predicted using VSEPR theory. Therefore, in polyatomic molecules (i.e., molecules made up of three or more atoms), the net dipole moment is the vector sum of all bond dipole moments present in the molecule. On the other hand, the molecular dipole moment (also referred to as the net dipole moment) is a measure of the polarity of the molecule as a whole. A bond dipole moment is a measure of the polarity of a chemical bond between two atoms in a molecule. It is important to note the distinction between bond dipole moments and molecular dipole moments. The arrow represents the shift of electron density in the molecule it points away from the less electronegative (and more positively-charged) hydrogen atom and towards the more electronegative (and more negatively-charged) fluorine atom. Shown below is an illustration of the dipole moment present in a molecule of hydrofluoric acid. Since they have both a magnitude and direction, dipole moments are considered to be a vector quantity. Greater differences in electronegativity will consequently lead to larger dipole moments. A dipole moment is a result of unequal electron distribution, which occurs due to differences in electronegativity between chemically-bonded atoms (since electrons tend to move towards more electronegative atoms). 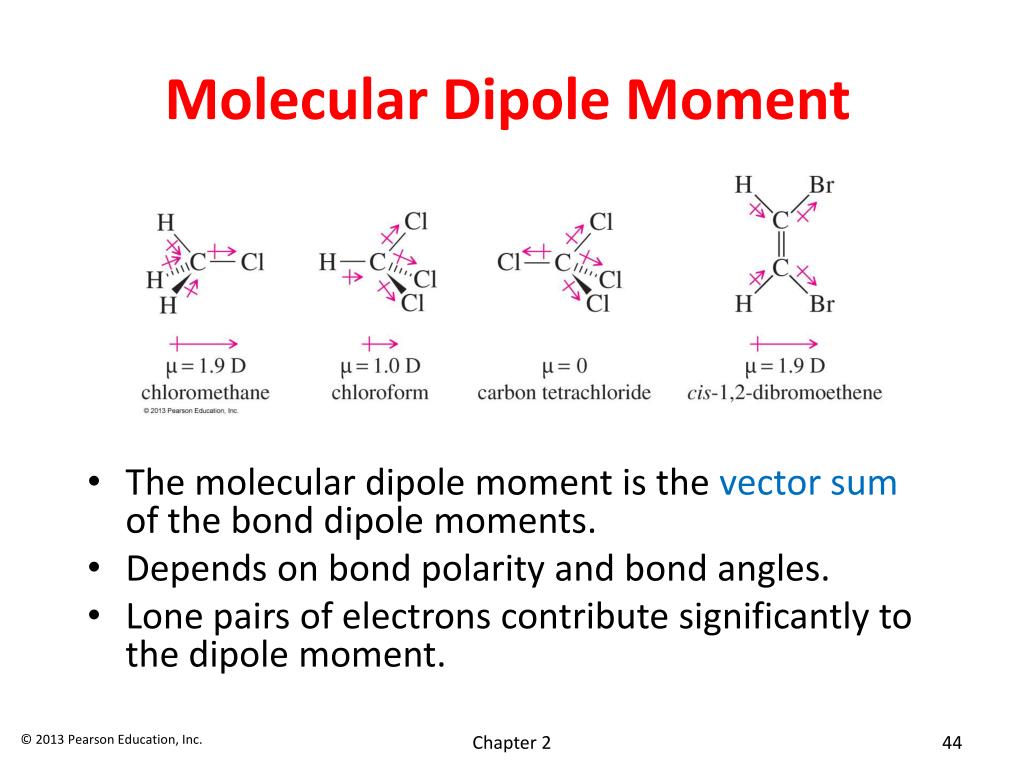
Topics Covered in Other Articlesĭipole moments occur in any system in which there is a separation of positive and negative electrical charges therefore, they can be found in both ionic and covalent bonds. Lastly, we provide several examples that demonstrate the importance of molecular geometry in determining net dipole moments. 
Then, we discuss the formula used to calculate dipole moments and explain their classification as a vector (rather than scalar) quantity. First, we begin with a basic introduction to the presence of dipole moments in chemical bonds and molecules. In this tutorial, you will learn all about dipole moments. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
